Healthcare labs deal with rules every day. They need to track quality checks, protect patient data, monitor equipment, and keep records ready for inspection. That is a lot to manage, especially when the work is still done with paper logs, spreadsheets, and manual reminders.
That is where healthcare automated lab compliance comes in. It helps healthcare laboratories use connected systems, smart alerts, and digital workflows to stay organized, reduce manual mistakes, and keep healthcar ecompliance tasks running in real time.
In this guide, you will learn what automated lab compliance means in healthcare, why it matters, how it works, and how healthcare IT support helps make it possible.
What Is Automated Lab Compliance in Healthcare?
Automated lab compliance in healthcare means using software, connected tools, and digital workflows to help a lab meet rules and standards without depending on manual processes all the time. Instead of checking everything by hand, the system helps track, record, and alert staff when something needs attention.
In simple words, it helps a lab stay ready. Quality control records, temperature logs, equipment checks, staff training records, and audit reports can all be handled through one connected setup. This makes daily work easier and helps the lab stay prepared for inspections.
Importance of Automated Lab Compliance in Healthcare

Healthcare labs compliance is not just about passing an inspection. It also affects patient safety, testing accuracy, data protection, and daily operations. When something is missed, the impact can go far beyond paperwork.
A missed temperature issue can affect stored specimens. An incomplete quality log can create problems during a review. A weak system for handling patient data can create privacy risks. Automation helps reduce these gaps by making compliance work more consistent and easier to manage.
What Does Lab Compliance Mean in Healthcare?
Lab compliance means following the rules that control how a healthcare laboratory works. These rules cover testing, quality control, equipment maintenance, staff training, documentation, and data security.
A compliant lab should be able to prove that it performs tests correctly, protects patient information, trains its staff, tracks its equipment, and keeps clear records. It is not just a box to check. It is part of running a safe and reliable laboratory.
5 Key Rules and Standards Healthcare Labs Must Follow
Healthcare labs work under several important rules. Each one plays a different role, and together they shape how lab compliance works.
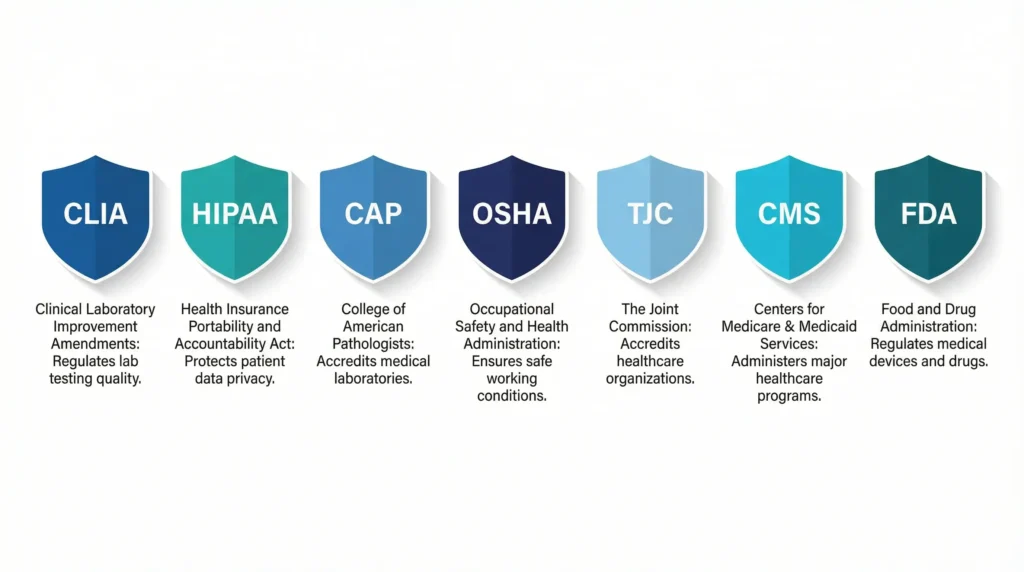
CLIA
CLIA stands for Clinical Laboratory Improvement Amendments. It sets quality standards for labs that test human samples. This includes quality control, test accuracy, staff qualifications, and proper documentation.
For most labs, CLIA is one of the main compliance foundations. If a lab does not meet CLIA requirements, it can face serious operational and regulatory problems.
HIPAA
HIPAA stands for the Health Insurance Portability and Accountability Act. It focuses on protecting patient health information. For labs, that means securing the systems that store, access, or share patient data.
This includes access control, audit logs, secure servers, and protected digital records. In other words, lab compliance is not only about testing. It is also about keeping patient data safe.
CAP
CAP stands for the College of American Pathologists. CAP accreditation adds another layer of quality and inspection readiness for medical laboratories. It expects labs to document what they do, follow their procedures, and keep complete records.
That is one reason automation helps so much. It makes it easier to store, track, and retrieve the records inspectors want to review.
OSHA
OSHA stands for the Occupational Safety and Health Administration. It focuses on workplace safety. In a lab, that includes exposure risks, staff safety practices, and required training.
Automated compliance tools can help track training, incident reports, and safety-related records so that important details do not get missed.
TJC
TJC stands for The Joint Commission. It accredits healthcare organizations and looks at how safely and effectively they operate. For hospital-based labs, TJC standards affect procedures, documentation, quality control, and clinical workflow integration.
This is where 24/7 managed IT services, 24/7 IT support, network monitoring, and infrastructure management help. They keep lab systems connected, secure, and aligned with the broader hospital IT environment, which supports TJC survey readiness.
CMS
CMS stands for the Centers for Medicare & Medicaid Services. It manages major healthcare programs and also plays a key role in lab oversight through CLIA-related regulation and certification.
For labs, CMS matters because compliance issues can affect certification, operations, and reimbursement in some healthcare settings.
FDA
FDA stands for the Food and Drug Administration. It regulates medical devices, drugs, and certain systems used in healthcare and laboratory environments.
When labs use digital systems, instruments, or regulated electronic records, FDA-related requirements can become part of the compliance picture.
Why Manual Lab Compliance Is Hard to Manage
Manual compliance may seem manageable at first. But as the lab grows, it often turns into a slow and messy process. Staff end up spending too much time checking logs, updating files, chasing signatures, and searching for records.
The bigger problem is that manual work creates blind spots. A missed entry, a delayed check, or a lost record can turn into a real issue later. By the time someone notices, the damage may already be done. That is why many labs are moving away from paper-heavy compliance systems.
6 Common Problems With Manual Healthcare Compliance
There are a few reasons manual compliance keeps breaking down in real healthcare settings. These problems show up again and again.

Human Error in Documentation
When staff manually enter QC results, temperature readings, and maintenance records, transcription errors, omissions, and retroactive entries are inevitable. These documentation gaps become deficiency citations during CAP, CLIA, or OSHA inspections and are among the most frequently cited reasons for corrective action.
No Real-Time Visibility
In a manual system, compliance failures are discovered after the fact, whether during a weekly review, a supervisor’s audit, or an inspector’s visit.
A freezer drifts out of the temperature range on Friday evening, and nobody knows until Monday morning. By then, specimens and reagents may be compromised.
Remote monitoring and alerting replace this reactive model with continuous, automated surveillance that sends instant notifications the moment a parameter drifts out of range, day or night.
Audit Preparation Consumes Weeks
Under manual compliance, assembling documentation for a CAP or CLIA inspection can consume hundreds of staff hours, including locating records across multiple locations, checking for completeness, and printing and organizing binders. That time is taken directly from patient care and laboratory operations.
Silos Destroy the Full Picture
QC records are in one spreadsheet. Equipment logs are in another. Training certificates sit in a shared folder that no one consistently maintains. There is no single source of truth, and there is no way to assess your actual compliance status without manually aggregating data from every silo.
Staff Turnover Creates Blind Spots
When a compliance officer or senior lab manager leaves, the institutional knowledge of how compliance is managed disappears with them. Manual systems have no mechanism for capturing procedures, decision logic, or compliance history in a structured, searchable format
Volume Growth Breaks Manual Systems
Post-pandemic laboratory testing volumes have increased dramatically. More tests mean more QC events, more equipment calibration deadlines, more proficiency testing cycles, and more personnel training records. Manual systems do not scale under this volume. They produce more errors, more gaps, and more regulatory exposure the harder they are pushed.
IT Infrastructure Behind Automated Lab Compliance
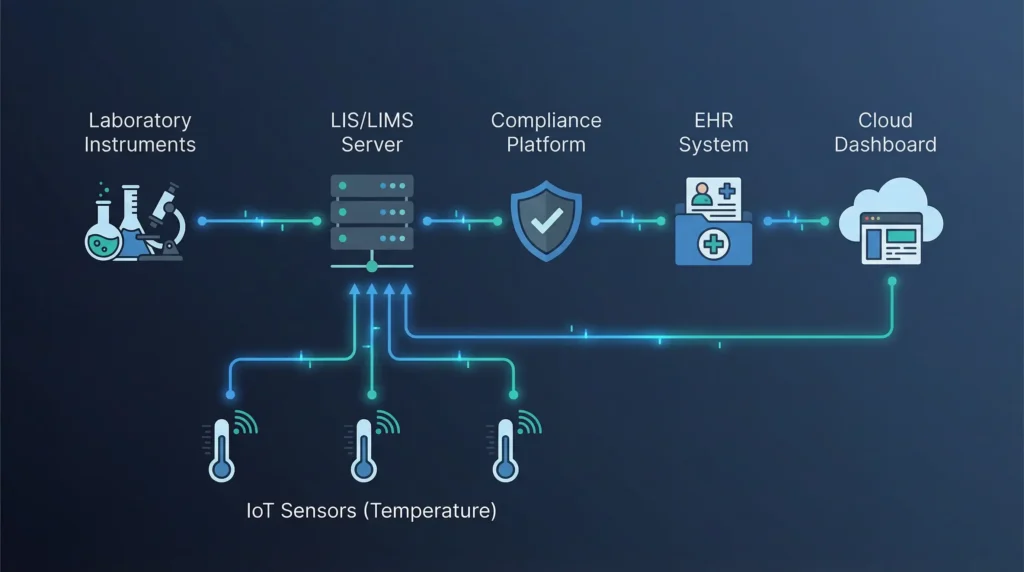
Automated lab compliance in healthcare is not just about software. It depends on a strong healthcare IT infrastructure that keeps systems connected, secure, and always running. Without the right setup, even the best compliance tools can fail.
In simple words, your compliance system is only as strong as the IT systems behind it. That includes network reliability, EHR and LIS integration, server security, data protection, and continuous monitoring.
Network Reliability Is Non-Negotiable
Automated lab compliance depends on systems sharing data in real time. IoT temperature-monitoring devices, lab instruments, LIS systems, and compliance dashboards all require a stable network to function properly.
If the network goes down, it does not just stop work. It can lead to missing compliance logs, audit gaps, and data loss, which can become serious issues during inspections.
That is why network monitoring and management services are important. They help track performance, detect issues early, and keep compliance data flowing without interruption.
EHR and LIS Integration
In healthcare labs, data is stored in different systems. The Laboratory Information System (LIS) handles test results and quality control data, while the Electronic Health Record (EHR) stores patient information and orders.
Without proper integration, staff have to move data manually between systems. This increases errors and slows down compliance work.
With EHR integration services and custom HL7 and FHIR interfaces, data moves automatically between LIS, EHR, and compliance platforms. This creates a connected healthcare system with accurate records and a clear audit trail.
Secured, HIPAA-Compliant Servers
Compliance data is sensitive. It includes patient-related data, QC logs, temperature records, staff information, and audit trails. This data must follow HIPAA security rules.
That is why labs need secure servers, server security, server hardening, data encryption, access control, and user authentication. These measures protect data from unauthorized access and cyber threats.
A strong healthcare IT security setup ensures your compliance system stays safe, reliable, and aligned with HIPAA requirements.
Proactive Server Monitoring for Zero Downtime
Automated lab compliance systems must run all the time. If a server goes offline, it can create gaps in monitoring and missing compliance records, which inspectors can flag.
Proactive server monitoring helps detect performance issues, hardware problems, and system failures early. It ensures high uptime, system stability, and continuous compliance tracking.
Backup and Disaster Recovery
Healthcare labs must store compliance records for long periods due to CLIA and HIPAA requirements. Losing this data can lead to serious legal and operational risks.
That is why backup solutions and disaster recovery management are critical. They protect data, ensure recovery after system failure or cyberattack, and maintain data integrity and compliance continuity.
Process Automation for Compliance Workflows
Many compliance tasks are repetitive. These include QC reporting, training reminders, calibration tracking, and CAPA (Corrective and Preventive Action) workflows.
With process automation and workflow automation tools, these tasks can run automatically. This reduces manual work, improves accuracy, and ensures nothing gets missed.
Using healthcare automation systems helps labs improve efficiency while maintaining consistent compliance.
How Healthcare Automated Lab Compliance Works
Automated lab compliance works by connecting systems, tools, and workflows so they can monitor activity and keep records without so much manual effort. Instead of relying on memory and paper, the system handles routine tracking in the background.
For example, a connected temperature monitor can send an alert the moment a storage unit moves out of range. A digital training system can remind managers when a competency review is due. A compliance dashboard can show open issues, completed tasks, and missing records in one place.
Main Parts of an Automated Lab Compliance System
A strong automated compliance setup usually includes several connected functions. Each one supports a part of the lab’s daily compliance work.

Real-Time Quality Control Monitoring
Quality control checks need to be tracked and reviewed carefully. Automation helps capture QC activity faster and can alert staff when something falls outside expected limits.
This improves visibility and reduces the chance of missing a problem before test results are affected.
24/7 Temperature and Environmental Monitoring
Labs often need to monitor refrigerators, freezers, incubators, and storage areas. Automated sensors can watch these conditions around the clock.
Instead of waiting for someone to notice an issue later, alerts can be sent right away so staff can respond quickly.
Equipment Calibration and Maintenance Tracking
Lab equipment needs regular checks, maintenance, and calibration. Automated systems help track due dates, service history, and overdue tasks.
That makes it easier to keep equipment ready for use and to show proof during an inspection.
Staff Training and Competency Records
Training records are part of compliance, too. Labs need to know who completed what, when it expires, and what still needs attention.
Automation helps keep these records organized and easier to review.
Policy and SOP Management
Policies and procedures change over time. Automated systems can help labs manage the latest versions, track staff acknowledgment, and avoid confusion caused by outdated documents.
Proficiency Testing (PT) Tracking
Proficiency testing helps labs show that their test results are accurate and consistent over time. With automated PT tracking, labs can manage deadlines, record results, and stay prepared for CLIA reviews without relying on manual work.
CAPA Workflow Management
CAPA workflow management helps labs take the right action when a compliance issue occurs. It keeps every step organized, from identifying the problem to fixing it and preventing it from happening again.
Audit Reporting
One of the biggest benefits of automation is faster reporting. Instead of gathering records by hand, teams can pull digital reports when needed.
That saves time and reduces stress during audits and inspections.
Role of IT in Automated Lab Compliance
This is the part many labs overlook. Automated compliance only works well when the IT side is strong. If the network is unstable, the server is weak, or the systems do not connect properly, the compliance process also becomes weak.
That is why automated lab compliance is not only a software decision. It is also an IT infrastructure decision. The systems behind the lab must be secure, connected, and monitored so compliance tools can keep running without interruption.
What Healthcare IT Support Helps With
Healthcare IT support helps labs keep the technology behind compliance stable and secure. It supports the systems that carry the data, store the records, and send the alerts.
That may include:
- secure servers
- EHR and LIS integration
- network monitoring
- backup and disaster recovery
- remote monitoring tools
- access control and data protection
- Ongoing technical support
When these parts are managed well, the compliance system becomes more reliable.
Key Benefits for Healthcare Labs and IT Teams

Automated lab compliance does more than help labs follow rules. It helps healthcare labs and IT teams reduce risk, save time, improve accuracy, and keep systems and records ready for inspections at all times.
Continuous Inspection Readiness
Rather than preparing for inspections, your lab is always prepared. The cost of that shift, measured in reduced staff stress, faster inspection completion, and fewer citations, is high.
Platforms serving labs with automated compliance report inspection preparation time dropping from two full working days to just 6 to 8 hours.
Up to 30% Reduction in Compliance Costs
Industry data shows automated compliance tools reduce compliance-related costs by up to 30% through improved efficiency, fewer regulatory violations, and reduced manual labor.
When you factor in the avoided cost of failed inspections, corrective action programs, and potential penalties, the ROI case for automation is overwhelming.
Reliable and Accurate Patient Test Results
When QC is monitored in real time, temperature excursions are caught within minutes, equipment is maintained on validated schedules, and staff are trained and assessed continuously, the laboratory produces more accurate and more reliable results. Automated compliance is fundamentally a patient safety program.
Staff Freed from Compliance Busywork
Every hour a laboratory professional spends manually logging compliance data, filing paper records, or assembling audit binders is an hour not spent on patient-facing work.
Automation reclaims that time, and with proactive IT support ensuring systems stay online and performant, staff can trust their tools rather than work around them.
Multi-Site and High-Volume Scalability
Multi-site laboratory networks benefit enormously from centralized automated compliance. A single dashboard provides real-time compliance visibility across every location, which is something categorically impossible with paper-based systems.
Enterprise operations management brings this multi-site coordination to laboratory IT operations, ensuring consistent compliance standards are maintained organization-wide.
Financial Protection from Growing Penalties
With OCR penalties for willful HIPAA neglect exceeding $71,000 per violation and the average healthcare data breach costing nearly $11 million, the financial risk of manual compliance is no longer theoretical.
SLA management gives laboratory IT operations a defined, measurable service-level framework, ensuring compliance infrastructure delivers the reliability that regulatory obligations require.
Defensible Audit Trails Under 21 CFR Part 11
Electronic records with validated audit trails, electronic signatures, and user access controls do not just satisfy inspectors. They protect laboratories in the event of disputed results, legal proceedings, or payer audits.
Data quality and validation services ensure that the integrity of the data moving through your compliance infrastructure meets the evidentiary standards that regulators and courts expect.
Manual vs Automated Healthcare Compliance

Manual compliance takes more time and leaves more room for errors. Automated compliance keeps records organized, improves visibility, and helps labs respond faster.
| Compliance Area | Manual Compliance | Automated Lab Compliance |
| QC Monitoring | Manual entry, periodic review | Real-time instrument capture with instant alerts |
| Temperature Monitoring | Staff check-ins 2 to 3 times per day | 24/7 IoT sensor monitoring with immediate SMS and email alerts |
| Equipment Calibration | Paper logs, memory-dependent scheduling | Automated reminders, digital records, and locked equipment alerts |
| Staff Training Records | Spreadsheets and shared drives | Centralized tracking with auto-expiration alerts and e-signature acknowledgment |
| Policy Management | Paper binders with ad-hoc review cycles | Version-controlled repository with automated review scheduling |
| Proficiency Testing | Manual tracking with frequent missed deadlines | Automated tracking, performance monitoring, and CAPA triggers |
| CAPA Management | Informal and inconsistent follow-through | Structured workflow with assigned deadlines and escalation alerts |
| Audit Preparation | Weeks of document gathering | Real-time dashboards with on-demand reports generated in minutes |
| Data Integrity | Vulnerable to human error and retroactive entry | Immutable audit trails with e-signatures and 21 CFR Part 11 compliance |
| Multi-Site Visibility | Impossible to centralize | Single dashboard covering all locations in real time |
| IT Infrastructure Need | Minimal, which is a core weakness | Requires reliable networks, secure servers, and integrated systems |
| IT Support Model | Reactive break/fix | Proactive managed IT with 24/7 support |
| Inspection Outcome | Reactive and anxiety-driven | Confident, evidence-based, and fully audit-ready |
How MediSure Solution Supports Healthcare Labs
Automated compliance does not work well on weak systems. If the network goes down, the records stop syncing, or the server fails, the lab can end up with gaps in its compliance process.
That is where MediSure Solution helps. MediSure supports healthcare organizations with the IT foundation needed for automated lab compliance. This includes healthcare IT support, monitoring, security, integrations, backup planning, and infrastructure support for medical laboratories and other healthcare environments.
Steps to Start Automated Lab Compliance
Labs do not need to change everything overnight. The best approach is to start with a clear plan and fix the biggest gaps first.
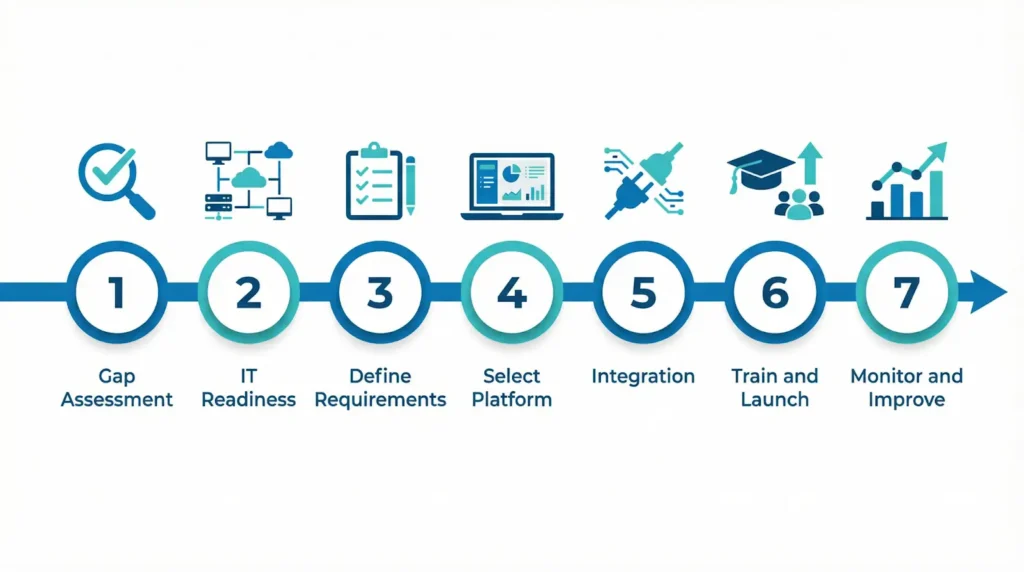
Step 1: Review Your Current Process
Look at how your lab handles QC, equipment tracking, temperature logs, staff training, and audit preparation today. Find what is still manual and where the risks are.
Step 2: Check Your IT Readiness
Review your network, servers, integrations, backup systems, and security controls. If the foundation is weak, automation will struggle too.
Step 3: Choose the Right Tools
Pick systems that fit your lab’s workflow and can connect with your current setup. The goal is to reduce silos, not create new ones.
Step 4: Train Your Team
Even a good system can fail if people do not know how to use it. Staff need training, clear roles, and a simple process to follow.
Step 5: Keep Improving
Once the system is live, keep tracking results. Look for repeated issues, missed alerts, or workflow gaps and improve them over time.
How to Choose the Right Healthcare IT Partner for Lab Compliance
Not every IT partner understands healthcare regulatory requirements. When evaluating a healthcare IT support provider for laboratory compliance infrastructure, apply this framework:
| Evaluation Criteria | Why It Matters |
| Healthcare-specific experience | Generic MSPs do not understand HIPAA, CLIA, or 21 CFR Part 11, and that gap shows up in your audit trail |
| 24/7 availability | Compliance monitoring runs at 3 AM and your IT support must too |
| EHR integration capability | Without LIS/EHR integration, compliance data silos remain intact |
| HIPAA compliance program | Your IT partner handles PHI and needs BAAs with documented HIPAA controls |
| Proactive support model | Reactive break/fix is not adequate for compliance-critical infrastructure |
| SLA transparency | Defined response times and uptime guarantees are compliance infrastructure requirements |
| Laboratory industry knowledge | Understanding CAP, CLIA, and laboratory workflows changes how IT problems are prioritized and resolved |
MediSure Solution was built specifically for healthcare organizations. With 3-plus years of dedicated healthcare IT experience, 99.9% SLA compliance, over 100 EHR integrations, and an average response time of under one minute, MediSure provides the IT support infrastructure that laboratory compliance automation requires.
Final Thoughts
Automated lab compliance in healthcare helps labs move away from scattered manual work and toward a more organized, connected, and reliable way of staying compliant. It supports daily operations, strengthens documentation, and helps labs respond faster when something goes wrong.
Most of all, it makes compliance easier to manage in the real world. And in healthcare, where the stakes are high, that matters a lot.
If you want, I can now turn this into a full final blog with:
meta title, meta description, FAQ schema content, internal links, and a strong MediSure CTA section.
Frequently Asked Questions:
What is automated lab compliance in healthcare, and how does it work?
Automated lab compliance means using digital systems to handle lab rules and checks automatically. It tracks things like test quality, temperature, and records in real time, so staff don’t have to manage everything by hand.
Why do healthcare labs need automated compliance systems?
Labs use automated compliance systems to avoid mistakes and save time. It helps keep records complete, sends alerts when something goes wrong, and makes it easier to pass inspections.
How does automated lab compliance improve patient safety and accuracy?
It improves safety by catching problems early, like failed tests or wrong storage temperatures. This helps labs fix issues quickly and ensures patients get accurate test results.
What are the benefits of automated lab compliance for hospitals and labs?
It reduces paperwork, saves staff time, keeps records organized, and makes audits easier. It also helps labs stay ready for inspections without last-minute stress.
How does healthcare IT support help with lab compliance automation?
Healthcare IT support keeps systems running, secure, and connected. It helps manage servers, networks, and data so the compliance system works without interruptions.
What systems are required for automated lab compliance in healthcare?
Labs need systems like lab software (LIS), patient record systems (EHR), monitoring devices, and secure IT systems. These work together to track data, send alerts, and store records safely.


